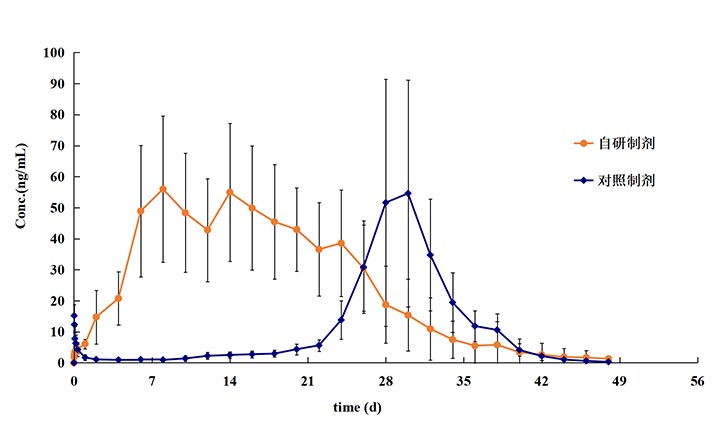
-
Long acting sustained-release microspheres
Long-acting injection microsphere technology is a safe and efficient drug release system, and biodegradable materials as carriers have been approved by the FDA and widely used. According to different drug properties and delivery requirements, drugs can be uniformly or phase separated and distributed in microspheres, effectively controlling the concentration fluctuations of drugs in the blood. This technology can be designed into sustained-release formulations ranging from weeks to months as needed. The applicable drugs include anticancer drugs, painkillers, hypoglycemic drugs, etc. While achieving therapeutic goals, it can also improve the bioavailability of drugs, thereby reducing drug dosage, lowering medication frequency, and minimizing toxic side effects.
-
Long acting sustained-release implant
Long acting sustained-release implant is a long-acting sustained-release drug system that is surgically implanted or needle injected into subcutaneous or other targeted sites. Long acting implants have the characteristics of achieving local or systemic administration, reducing dosage and minimizing side effects, avoiding first pass effects and gastrointestinal degradation, improving bioavailability, high drug loading, small volume, and prolonging drug action time. They are particularly suitable for long-term treatment of chronic and elderly diseases, and improving patient compliance. Implants can eliminate the phenomenon of drug concentration peaks and valleys caused by intermittent administration, and can continuously release drugs at a constant rate at specific sites of action to maintain stable therapeutic concentrations. They can achieve therapeutic effects with small doses.
-
In situ gel
In situ gel is a kind of drug repository that rapidly changes its physical form and forms a semi-solid gel state at the application site after injection in a relatively low viscosity liquid state. In situ gel has good histocompatibility, long retention time at the drug delivery site, and can also play the role of storing drugs and preventing drugs from being affected by the environment. According to different formation mechanisms, in-situ gel can be divided into temperature sensitive, pH sensitive and ion sensitive types. As a new drug dosage form, in situ gel is widely used in new drug delivery systems such as sustained release, controlled release and pulse release. In situ gel can be used for subcutaneous or intramuscular injection, skin, eye, nasal cavity, oral cavity, vagina, rectum and other routes of drug delivery.