Application Scope
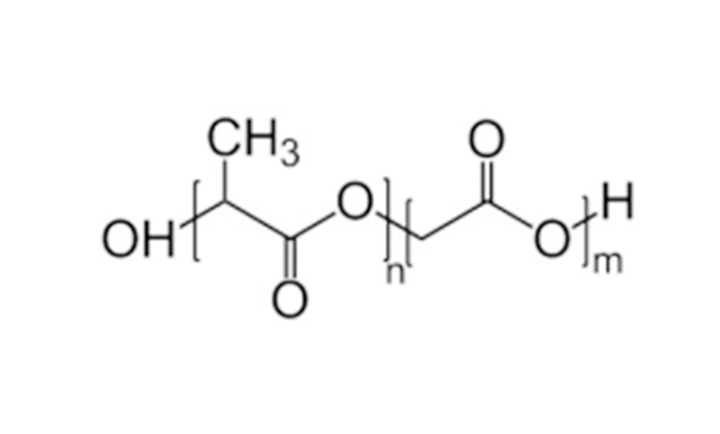
PLGA is an important class of biomedical polymer materials that combines the advantages of two polyester materials, polylactic acid (PLA) and polyglycolic acid (PGA), with good biocompatibility and degradability. It is widely used in the biomedical field, such as surgical sutures, fracture internal fixation materials, tissue repair materials, and drug controlled release systems. The main advantage of bioabsorbable polymers is that they are hydrolyzed and degraded into monomers, breaking down the components lactic acid and glycolic acid - both of which are absorbed by the body. The degradation degree of PLGA varies with different monomer ratios, and the higher the proportion of ethyl ester, the easier it is to degrade. PLGA has been approved for human use by drug review centers worldwide, including the United States, European Union, China, Japan, and other countries and regions, and is safe and reliable.
Application Cases
Application cases of injection preparations
| Product name |
Manufacturer |
Indications |
Dosage form |
Polymer model |
| Risperidone microspheres for injection |
Janssen |
Schizophrenia |
Microspheres |
75/25 |
| Leuprorelin Acetate Microspheres For Injection |
Takeda |
Breast cancer |
Microspheres |
75/25 95/05 100/0 |
| Goserelin Acetate Sustained-Release Depot |
AstraZeneca |
Prostate cancer |
Subcutaneous implants |
50/50 95/05 |
| Dexamethasone intravitreal implant |
Allergan |
Macular edema |
Vitreous implant |
50/50 |
Medical Device Application Cases
Coronary drug-eluting stent: The stent material is 316 stainless steel, the polymer is PLGA, and the drug is rapamycin.
VICRYL surgical suture: PLGA9010, absorbed within 3 months.